 Plastic surgeons are well known for their ability to fix a nose or provide implants for breast, chin, or cheek augmentation. Nevertheless, when the bones of the face are injured and require reconstructive surgery, repairing the damage has been a daunting task for even the most seasoned plastic surgeon. They pore over CT and MRI scans, and physically examine the remaining facial bone structure, all in the hopes of remaking the damaged area look as it did before the injury.
Plastic surgeons are well known for their ability to fix a nose or provide implants for breast, chin, or cheek augmentation. Nevertheless, when the bones of the face are injured and require reconstructive surgery, repairing the damage has been a daunting task for even the most seasoned plastic surgeon. They pore over CT and MRI scans, and physically examine the remaining facial bone structure, all in the hopes of remaking the damaged area look as it did before the injury.
Saving Face
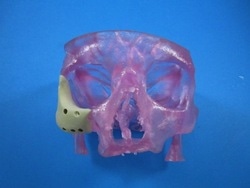
Today things have been changing quite a bit in the world of facial reconstructive surgery. The company Oxford Performance Materials (OPM) has just been issued FDA approval for the first and only 3D-printed implantable device for facial reconstructive surgery. This approval came on the heels of another in February for the company’s OsteoFab™ Patient Specific Cranial Device (OPSCD). Both devices are constructed using polyether ketone ketone (PEKK) a high-performance thermoplastic.
The ability to 3D print these implants makes them totally customizable for each patient. Using photographs, MRI scans, and other images, the implant can be tailor-made to match and effectively fit right into place. 3D printing will lower the cost of the implants and speed up the turnaround time it takes to make them. Once the surgeon is happy with the design, the implant can be printed and ready for implantation in hours compared to days or weeks. Moreover with specific customization, the hope is to decrease surgical time thereby lowering exposure to infection, as well as patient cost.
Scott DeFelice, chief executive officer and chairman of Oxford Performance Materials said:
Until now, a technology did not exist that could treat the highly complex anatomy of these demanding cases. With the clearance of our 3D printed facial device, we now have the ability to treat these extremely complex cases in a highly effective and economical way, printing patient-specific maxillofacial implants from individualized MRI or CT digital image files from the surgeon. This is a classic example of a paradigm shift in which technology advances to meet both the patient’s needs and the cost realities of the overall healthcare system.
Biocompatible Material
The company uses a technique of combining laser sintering additive and its proprietary PEKK powder preparation, OXPEKK®. The resulting material is biocompatible, so it has no adverse effect on the surrounding tissue. When implanted in the body, it is mechanically similar to bone and is osteoconductive as it proliferates the growth of bone tissue. It’s also radiolucent, making it almost transparent under radiation such as an X-ray or fluoroscope.
Improved Care, Reduced Cost
When faces require reconstructive surgery, whether from trauma or disease, surgeons work painstakingly to help these patients regain their appearance. A 3D-printed fully customizable facial implant for reconstructive surgery will help the surgeon achieve the goal of providing a safe way to restore the patient, keeping costs and post-surgical complications down as well.
This is a very exciting time in medicine, as we are seeing more and more being achieved through the wonders of 3D printing, which is becoming a game changer in the health-care industry.
Image by Oxford Performance Materials.
Source: “Oxford Performance Materials Receives FDA Clearance for 3D Printed OsteoFab® Patient-Specific Facial Device,” by OPM, www.oxfordpm.com, August 19, 2014.
Source: “FDA Approves First 3D Printed Facial Implants,” by Hannah Rose Mendoza, www.3dprint.com, August 20, 2014.
Source: “Oxford Performance Gets FDA Approval for Facial Implant Material,” by Todd Halterman, www.3dprinterworld.com, September 8, 2014.
Source: “First FDA Clearance for 3D Printed Polymeric Implants for Facial Reconstruction,” by editors, www.medgadget.com, August 28, 2014.
